Summer 2022 Peach Picking Adventures (Branstool Orchards) Pictured from left to right: Zhihao Cui, Chris Bernard, Sung Gu Cho, Melissa Marx, Dr. Anne Co, Maddie Palardy, Jiseon Hwang, Modeste Tegomoh, Dave Wood, and Albert
Ph.D Defense and Successful Candidacy Exam Celebration November 2023 (Marcella’s Polaris): Pictured from left to right: Qingmin Xu, Dr. Anne Co, Dr. Kug-Seung Lee, Ariel Mendoza, Dr. Zhihao Cui, Modeste Tegomoh, Dave Wood, Maddie Palardy, Dr. Sung Gu Cho, Albert Wang. Front row left to right: Dr. Melissa Marx, Sam Mendez, and Jiseon Hwang
Group Get Together Summer 2021 (from left to right): Chris Bernard, Sung Gu Cho, Julen Bascaran, Daniel Lyons, Anna Lisa, Dave Wood, Zhihao Cui, Modeste Tegomoh, (Back Row left to right) Ariel Mendoza, Ben Charnay, Aaron Charnay, Dr. José L. Lorié López, (Front Row left to right) Jiseon Hwang, Dr. Anne Co, and Melissa Marx
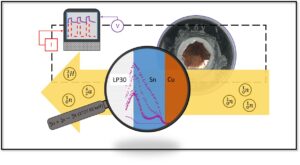
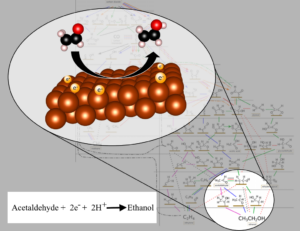
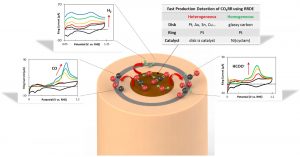
The primary focus of our research effort is in the fundamental studies of electrochemical reactions, electrocatalyst function and the design of new materials for furthering electrochemical technologies. Our lab is multidisciplinary, combining electrochemical, analytical, materials and physical chemistry techniques.
Main Research Goals
- Gain deeper fundamental understanding of electrocatalyst reactivity and selectivity
- Design, characterize, and evaluate new energy conversion and storage electrode materials
- Develop systematic methods of studying the mechanistic pathways of electrochemical reactions
- Utilize and develop in-situ methods for detecting reaction intermediates to infer reaction pathways